In the legal arena of pharmaceutical lawsuits, the case of Suboxone presents a fascinating evolution from significant class action lawsuits addressing antitrust and drug pricing disputes to the more recent surge in personal injury claims centered on dental issues.
This article discusses the nature of these legal battles, highlighting the complexities and outcomes of the earlier class action lawsuits against Suboxone’s manufacturers for anti-competitive practices and juxtaposing them with the ongoing personal injury lawsuits that allege severe dental injuries from the drug’s use.
As we explore these distinct legal chapters, we uncover the multifaceted implications of Suboxone litigation, shedding light on corporate accountability in pharmaceutical marketing and the pressing concerns over drug safety and consumer health.
The Suboxone Class Actions Over Drug Pricing
The Suboxone class action lawsuit was a significant legal matter involving allegations of antitrust violations by the makers of Suboxone, a prescription drug used to treat opioid addiction.
The Federal Trade Commission (FTC) sued the makers of Suboxone, alleging that they were preventing patients from choosing lower-priced generic versions of the drug. To settle these FTC charges, the companies agreed to pay $60 million to consumers.
In addition to the FTC lawsuit, a separate $30 million class action lawsuit was settled over similar antitrust allegations. This lawsuit claimed that the actions of the companies led to consumers and patients overpaying for Suboxone and its “AB-rated” generic equivalent medications.
The FTC later distributed payments totaling more than $59 million to consumers who were victims of the alleged deceptive scheme by Reckitt Benckiser Group and Indivior, Inc. This scheme was aimed at thwarting lower-priced generic competition with the branded drug Suboxone. A total of 51,875 payments were sent, with an average payment amount of $1,139.
Moreover, the company that makes Suboxone reached a $102.5 million settlement of antitrust claims with 41 states and the District of Columbia. This settlement averted a trial that was scheduled to start later that year. The attorneys general of these states had sued Indivior, claiming the company schemed to keep generic competitors off the market.
Lastly, Indivior also agreed to pay $30 million to settle a class action lawsuit filed in a U.S. court by health plans. These health plans accused the drugmaker of illegally suppressing competition for Suboxone.
The Suboxone Personal Injury Lawsuits Over Dental Issues
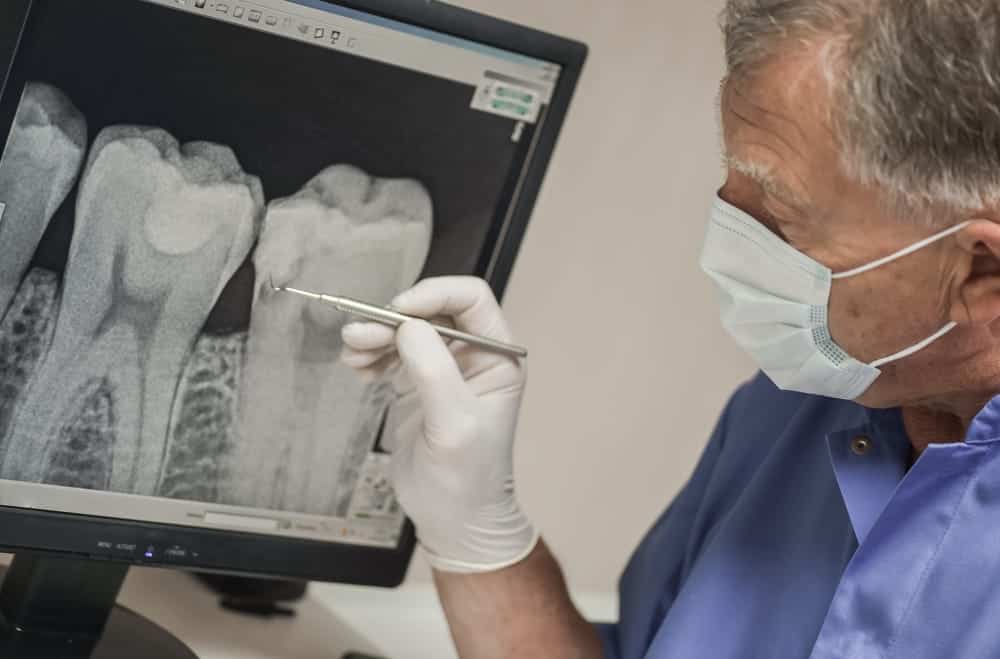
The recent personal injury lawsuits against the makers of Suboxone primarily focus on dental problems experienced by users of the drug.
Suboxone, a medication approved to treat opioid disorder, has been alleged to cause serious dental issues such as:
- tooth decay
- oral infections
- cavities
- tooth and bone loss
The lawsuits claim that the manufacturer, Indivior, failed to adequately warn about these risks, and plaintiffs are seeking compensation for the damages they have suffered.
As of November 2023, these lawsuits concerning Suboxone-related tooth decay were still in the early stages.
Lawyers are continuing to accept and investigate new cases, but there have been no court-approved, publicly announced settlements for dental problems related to Suboxone, nor have any trials been scheduled.
In November 2023, plaintiffs’ lawyers requested the U.S. Judicial Panel on Multidistrict Litigation to centralize all federal Suboxone lawsuits into a single multidistrict litigation.
This move was likely aimed at streamlining the legal process for the numerous cases being filed. In the same month, 14 new lawsuits specifically addressing Suboxone-induced tooth decay were filed against Indivior in federal courts, with the Northern District of Ohio seeing the highest number of these pending cases.
One notable case was filed in September 2023 by David Sorensen, who claimed that his use of Suboxone resulted in permanent dental damage and required significant dental work. His lawsuit targeted Indivior, Reckitt Benckiser, and other defendants.
These lawsuits followed the January 2022 announcement by the FDA that medicines containing buprenorphine, which dissolve in the mouth like Suboxone, could cause dental problems.
This revelation led to the addition of a new warning to the drug’s prescribing information and patient medication guide.
Please note: Due to statute of limitations deadlines that have now passed in most states, many attorneys are no longer accepting new Suboxone tooth decay cases. If you believe you may have a claim, consult with a qualified attorney in your state as soon as possible to determine whether your case is still eligible.